With complete biological definition and ensured reproducibility, recombinant antibodies from Absolute Antibody, a Vector Laboratories company, power researchers to study areas like neuroscience, infectious disease, and immunotherapy with confidence.
The majority of reagent monoclonal antibodies available today are generated from hybridomas, or immortal antibody-producing cells created by fusing a tumor cell with an antibody-producing B cell from an immunized animal. However, hybridomas suffer from limitations like batch-to-batch variability as a result of genetic drift and genetic instability that impacts expression. Additionally, more than 30% of hybridomas are not actually monoclonal (https://doi.org/10.1080/19420862.2018.1445456) and contain additional antibody genes, interfering with experimental outcomes.
In contrast, recombinant antibodies are manufactured using synthetic genes expressed in a defined in vitro cell line. They offer a variety of benefits compared to traditional hybridoma-produced antibodies, like:
- Ensured reproducibility: Recombinant antibodies are absolutely defined by the amino acid sequence, which means batch-to-batch reproducibility is guaranteed.
- 100% biological definition: Recombinant antibodies have no variation in heavy or light chains and perform the same vial to vial.
- High purity: Recombinant antibodies from Absolute Antibody, a Vector Laboratories company, are expressed in chemically defined, serum-free mammalian expression systems with low endotoxin levels.
- Additional antibody value: Recombinant production opens antibody engineering opportunities to expand experimental possibilities. Antibody engineering options include species and isotype swapping, fragment and multispecifics creation, chimerizing and humanizing antibodies, and Fc region silencing to reduce nonspecific staining and off-target toxicity in therapeutic applications.
- Animal-free manufacturing: Recombinant antibodies are manufactures in vitro using synthetic genes, eliminating animal welfare concerns associated with traditional monoclonal antibody manufacturing.
- Security: Recombinant antibodies begin at the sequence level, making them impervious to contamination, genetic drift, or accidental loss.
Recombinant antibodies from Absolute Antibody, a Vector Laboratories company, are highly specific research tools that grant scientists reproducibility, confidence in results and supply chain, and experimental flexibility in diverse fields.
How recombinant antibodies empower research across fields
With complete biological definition and ensured reproducibility, recombinant antibodies power researchers to study areas like neuroscience, infectious disease, and cancer and immunotherapy with confidence.
Neuroscience
Neuroscience research increasingly depends on tools that can resolve extreme cellular and molecular complexity, from defining thousands of distinct neuronal subtypes to tracking subtle protein changes across development and disease. Recombinant antibodies targeting well-established neuronal and glial markers like MAP-2 ([AA5 (AP18)]; Ab02896) and synaptophysin ([SP15]; Ab0030). Recent studies have leveraged these reagents in a variety of applications to unravel the complexity of neurobiology. For example, researchers used Absolute antibody’s anti-Ctip2 [25B6] recombinant antibody to distinguish layer- and region-specific neuronal and astrocyte populations
Another study (https://doi.org/10.1083/jcb.202303138) used the recombinant anti-polysialic acid clone 735 for immunofluorescence and western blot applications, finding that inhibiting polysialic acid’s post-translational modification of neural cell adhesion molecule 1 (NCAM-1) leads to improper formation and spacing of those specialized cells in the ventral conduction system of the heart (https://doi.org/10.1242/dev.199431).
In neurodegenerative disease research, recombinant antibodies enable the study of disease mechanisms like toxic protein aggregation, excitotoxic signaling, axonal degeneration, and neuroinflammation. In Alzheimer’s and Parkinson’s disease models, recombinant antibodies enable reproducible detection of targets like amyloid beta, tau, and polysialic acid. In ALS research, antibodies against misfolded SOD1, glutamate receptors, GFAP, neurofilament light chains, and inflammatory mediators such as IL-6 and HMGB1 have supported biomarker discovery, mechanistic studies, and preclinical evaluation of antibody-based interventions, linking molecular pathology to neuronal loss and functional decline.
Recombinant antibodies from Absolute Antibody are produced with full sequence definition to deliver batch-to-batch reproducibility, validated specificity, and flexible engineering into multiple species, isotypes, and formats. This recombinant-first approach supports consistent, high-confidence data generation across neuroscience, neurodegeneration, and ALS research, while enabling scientists to tailor antibodies precisely to their experimental needs.
Infectious Disease
Recombinant antibodies empower infectious disease research by providing sequence-defined, reproducible tools for studying viral genomes, replication intermediates, and host–pathogen interactions with high specificity. Antibodies against double-stranded RNA (dsRNA), a viral replication intermediate, support sensitive detection of viral replication across diverse virus families. In a recent study, the recombinant anti-dsRNA [J2] antibody (Ab01299) was used in immunofluorescent detection of sites of viral replication in order to study the role of CD4+ T cells in SARS-CoV-2 viral clearance.(https://doi.org/10.3389/fcimb.2025.1621226)
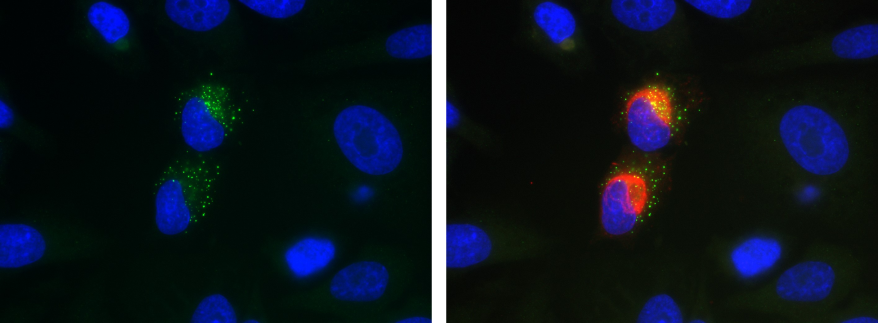
Additionally, engineered antibodies targeting key viral proteins such as the highly cited antibody anti-SARS-CoV-2 S glycoprotein clone CR3022, flavivirus group antigen, Nipah virus F and G glycoproteins, HIV antigens, and hepatitis surface proteins facilitate applications spanning immunofluorescence, ELISA, neutralization assays, and serological assay development.
With complete biological definition and flexible engineering options—including species switching, Fc silencing, and Fab or F(ab’)₂ fragment formats—recombinant antibodies enhance assay consistency, reduce background, and expand experimental design possibilities. This precision and adaptability strengthen studies of viral entry, replication, immune evasion, and inflammatory signaling, accelerating target validation, diagnostic development, and vaccine research across emerging and established infectious diseases.
Cancer and Immunotherapy
Recombinant antibodies are transforming immunotherapy and cancer research by giving scientists precise, biologically defined tools to dissect and manipulate immune checkpoints and tumor-associated targets in clinically relevant ways. Engineered antibodies against PD-1 (e.g., clone RMP1-14 in mouse IgG2a Fc Silent™ format, PD-L1 [10F.9G2], PD-L2 [TY25], CTLA-4, TIGIT [1B4], and more) enable detailed study of immune regulation, tumor immune evasion, and resistance mechanisms within the tumor microenvironment. Recombinant research-grade biosimilars such as anti-HER2 [4D5-8 (trastuzumab)] and anti-TNFα [cA2 (infliximab)] further support mechanistic and translational studies across breast, colorectal, prostate, and lung cancers, bridging preclinical modeling with clinically validated targets.
To meet the needs to cancer and immunotherapy research, recombinant antibody engineering can address complex problems. For example, our VivopureX™ antibodies are species-matched (mouse-anti-mouse), Fc-engineered, and produced at high purity with low endotoxin levels to optimize in vivo performance in mouse models. With options against clinically relevant targets, VivopureX™ antibodies provide optimized performance in vivo. Fc Silent™ variants allow true checkpoint blockade without unintended Fc receptor–mediated depletion, while IgG2a formats can enhance FcγR engagement where Treg depletion or myeloid activation is desired—critical for targets such as CTLA-4 and TIGIT.
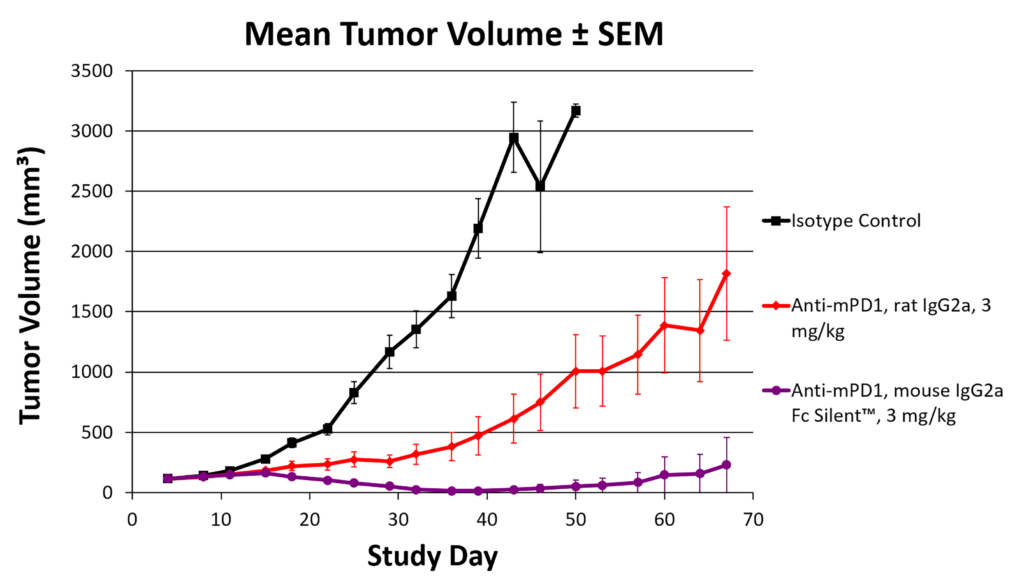
Data showing improved tumor reduction with Fc-silenced PD-1 mouse IgG2a antibodies compared to traditional rat formats (Figure 1) illustrate how recombinant engineering directly impacts experimental outcomes. Together, fully defined recombinant antibodies empower researchers to interrogate immune checkpoint biology and other key foci in immunotherapy research with reproducibility, flexibility, and security.
Conclusion
Recombinant antibodies address the limitations of hybridoma-derived reagents by providing sequence-defined, fully characterized tools with consistent performance across batches. Their biological definition, purity, and engineering flexibility support rigorous experimental design in complex research areas including neuroscience, infectious disease, and cancer immunotherapy.
By starting at the genetic sequence level, recombinant antibodies improve reproducibility, supply stability, and confidence in long-term research outcomes.
Explore the Absolute Antibody catalog to identify sequence-defined reagents and engineered formats suited to your specific research needs.
References
- Tan, C. X., Bindu, D. S., Hardin, E. J., Sakers, K., Baumert, R., Ramirez, J. J., Savage, J. T., & Eroglu, C. (2023). δ-Catenin controls astrocyte morphogenesis via layer-specific astrocyte–neuron cadherin interactions. The Journal Of Cell Biology, 222(11). https://doi.org/10.1083/jcb.202303138
- Elsharkawy, A., Dim, C., Ge, C., Patterson, L. D., Nabi, Z., & Kumar, M. (2025). SARS-CoV-2 XBB.1.5 infects wild-type C57BL/6 mice and induces a protective CD4+ T cell response required for viral clearance. Frontiers in Cellular And Infection Microbiology, 15, 1621226. https://doi.org/10.3389/fcimb.2025.1621226
- Delgado, C., Bu, L., Zhang, J., Liu, F., Sall, J., Liang, F., Furley, A. J., & Fishman, G. I. (2021). Neural cell adhesion molecule is required for ventricular conduction system development. Development, 148(11). https://doi.org/10.1242/dev.199431
- Bradbury, A. R. M., Trinklein, N. D., Thie, H., Wilkinson, I. C., Tandon, A. K., Anderson, S., Bladen, C. L., Jones, B., Aldred, S. F., Bestagno, M., Burrone, O., Maynard, J., Ferrara, F., Trimmer, J. S., Görnemann, J., Glanville, J., Wolf, P., Frenzel, A., Wong, J., . . . Dübel, S. (2018). When monoclonal antibodies are not monospecific: Hybridomas frequently express additional functional variable regions. mAbs, 10(4), 539–546. https://doi.org/10.1080/19420862.2018.1445456
Antibodies | Glycobiology | Immunodetection Reagents | Specialty Chemicals | Mounting Medium | Staining